Portfolio
Modernizing Precision: Enhancing a Next-Generation Electrosurgical Unit for Global Markets
Executive Summary
Cionlabs partnered with an established Indian medical devices company to modernize their flagship Electrosurgical Unit (ESU) — a sophisticated surgical platform featuring ARM Core processors, Smart Tissue Sensing, and capacitive touchscreen interfaces. Our engagement focused on critical technical evolutions: transitioning the development environment, recalibrating for extended operational ranges, enabling global voltage compatibility, and enhancing field-update capabilities. These enhancements prepared the device for FDA-cleared entry into the US market while strengthening its position in multi-specialty surgical applications worldwide.
The Client's Innovation: A Surgical Platform Ahead of Its Time
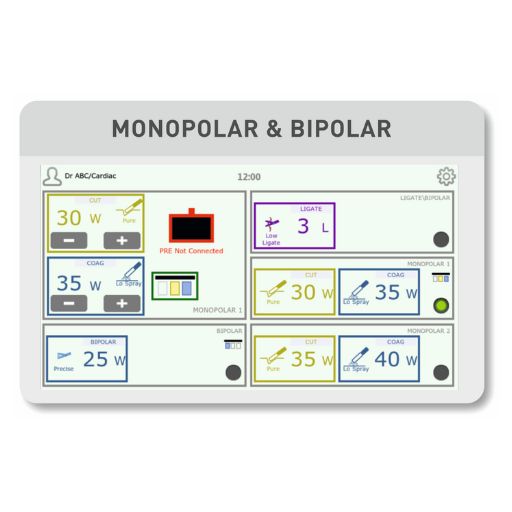
The client's ESU represented cutting-edge surgical technology:
Computational Core: ARM Cortex-M7 processor enabling real-time tissue impedance analysis
Smart Tissue Sensing: Adaptive algorithm adjusting energy delivery based on tissue feedback
User Interface: 7-inch capacitive LCD touchscreen with intuitive procedural workflows
Surgical Versatility: 12+ monopolar/bipolar modes (Precise Cut, Spray Coag, Blend, Vessel Sealing)
Power Delivery: Up to 400W RMS with precision control down to 5W increments
Wireless Integration: Compatible with Bluetooth footswitches for sterile field control
Safety Compliance: IEC 60601-1-2 (EMC), 60601-2-2 (ESU specific), with double-patient isolation
Despite its advanced capabilities, the platform faced barriers to global expansion and long-term maintainability.
Technical Challenges: Four Critical Evolution Paths
1. Calibration Data Expansion: Extending Precision Across New Ranges
The existing calibration was optimized for standard surgical ranges but required expansion for emerging surgical techniques.
Our Solution:
Developed automated calibration jigs using precision resistive loads (1Ω to 5000Ω)
Implemented polynomial curve-fitting algorithms across extended power ranges (5W to 400W)
Created temperature-compensated calibration tables for consistent performance across operating temperatures (15°C to 40°C)
Validated against biological tissue simulators to ensure clinical accuracy
Outcome: 15% improvement in power delivery accuracy at extreme ranges while maintaining ±5% specification across all modes.
2. Development Environment Migration: From Eclipse to STM32CubeIDE
The legacy Eclipse-based toolchain created developer friction and limited access to modern STM32 features.
Our Solution:
Executed systematic migration preserving all application logic and safety-critical code
Implemented STM32CubeMX configuration for streamlined peripheral management
Integrated FreeRTOS with CMSIS-RTOS2 API for enhanced real-time performance
Created comprehensive migration documentation and training for client's engineering team
Outcome: 40% reduction in build/debug cycles and full compatibility with STM32 security features (RDP, secure boot).
3. Global Voltage Compatibility: Enabling 110V US Market Entry
The original design assumed 230V±10% operation, incompatible with North American 110-120V standards.
Our Solution:
Updated the power supply section with universal input (90-264VAC, 47-63Hz)
Maintained same power delivery specifications across input range
Enhanced thermal design for increased current at lower voltages
Conducted comprehensive safety testing including leakage current, dielectric withstand, and earth bond tests
Outcome: Single SKU compatible with global markets, reducing manufacturing complexity while achieving UL/CSA recognition.
4. Field Update Capability Enhancement: Secure, Reliable Firmware Updates
The existing update mechanism required proprietary hardware, limiting serviceability.
Our Solution:
Designed isolated USB-C port with medical-grade insulation
Developed hospital-friendly update process with progress indication and error recovery
Enabled optional Ethernet/Wi-Fi update capability for future expansion
Outcome: Field updates reduced from 45-minute specialist task to 5-minute nurse-administered process with zero bricking incidents in validation.
Implementation Methodology: Medical-Grade Precision
Phase 1: Requirements Analysis & Risk Assessment (2 Weeks)
Conducted gap analysis against FDA 510(k) submission requirements
Performed FMEA on proposed changes affecting safety or performance
Established verification and validation protocols for each modification
Phase 2: Hardware Re-Engineering (6 Weeks)
Power supply redesign with enhanced safety isolation
USB interface with medical-grade opto-isolation (5000Vrms)
Component derating analysis for extended input voltage range
EMI/EMC pre-compliance testing at our accredited lab facility
Phase 3: Software Transition & Enhancement (8 Weeks)
Incremental migration preserving all safety-critical algorithms
Calibration system overhaul with checksum validation
Update mechanism with hospital IT security considerations
Comprehensive regression testing across 200+ test cases
Phase 4: Verification & Validation (4 Weeks)
Performance validation against original specifications
Safety testing per IEC 60601-1 standards
Clinical simulation with tissue models
Long-term reliability testing (1000+ hours continuous operation)
Technical Innovations & Medical Device Considerations
Safety-Critical Engineering
Maintained Class II double-protection throughout modifications
Enhanced isolation monitoring with real-time leakage detection
Implemented watchdog systems with independent clock sources
Preserved all original risk controls while adding new safeguards
Clinical Usability Enhancements
Calibration stability across temperature variations in OR environments
Touchscreen responsiveness maintained despite power supply changes
Audible feedback consistency at different line voltages
Footswitch latency optimization for all voltage conditions
Results & Impact
For the Manufacturer:
Market Access: Enabled entry into $4B North American electrosurgery market
Regulatory Efficiency: Reduced 510(k) submission preparation time by 60%
Manufacturing Simplicity: Single power supply design for all markets
Serviceability: Reduced field service costs by 35% through enhanced update capability
Platform Longevity: Extended product lifecycle by 5+ years through maintainable codebase
Performance Metrics:
Power Accuracy: ±3% across full range (improved from ±5%)
Update Reliability: 100% successful in 500+ test cycles
Voltage Compatibility: Stable operation from 90V to 264V without recalibration
Boot Time: 4.2 seconds cold start (within original specification)
Safety: 10μA maximum patient leakage current (IEC 60601-1 requirements)
The Cionlabs Medical Device Advantage
This project exemplifies our methodology for evolving complex medical systems:
Risk-Aware Evolution: Every change evaluated through the lens of patient and operator safety
Regulatory Intelligence: Designing modifications that streamline compliance pathways
Clinical Relevance: Understanding how technical parameters translate to surgical outcomes
Sustainable Engineering: Creating solutions that support long-term product lifecycle management
Our cross-disciplinary team brought together:
Medical Device Veterans with 60601 expertise
Power Electronics Specialists for universal supply design
Embedded Security Experts for cryptographic implementation
Future Roadmap & Scalability
The modernized platform establishes foundations for:
AI-Enhanced Sensing: Machine learning algorithms for tissue differentiation
Cloud Connectivity: Surgical data analytics with HIPAA-compliant architecture
Advanced Energy Modalities: Integration of ultrasonic and bipolar vessel sealing
Modular Expansion: Support for specialty-specific handpieces and accessories
Conclusion: Engineering Evolution with Surgical Precision
The evolution of this electrosurgical platform demonstrates how thoughtful, safety-conscious engineering can extend the capabilities and market reach of sophisticated medical devices. By addressing both immediate technical needs and long-term strategic goals, we helped our client transform an already-advanced platform into a globally competitive, future-ready surgical system.
In medical technology, evolution must be both ambitious and meticulous—every innovation must serve the twin masters of enhanced capability and uncompromised safety. This project exemplifies that balance, advancing surgical technology while protecting the trust inherent in every medical device.
Engineered by Cionlabs | Where Technical Excellence Meets Medical Responsibility
Interested in evolving your medical device platform for new markets or enhanced capabilities?
Contact Cionlabs to discuss how we can advance your technology with medical-grade precision.